
Hymovis FDA Approval Status
Hymovis is an FDA-approved, high-molecular-weight hyaluronic acid injection designed to treat knee osteoarthritis by improving joint lubrication and reducing pain.
All Products
B
M
R
S
U
W
Z
Find industry news, trends, and innovative products right here in our blog.
A
B
C
D
E
F
G
H
I
J
K
L
M
N
O
P
R
S
V
W
X

Hymovis is an FDA-approved, high-molecular-weight hyaluronic acid injection designed to treat knee osteoarthritis by improving joint lubrication and reducing pain.

Hymovis is an advanced hyaluronic acid-based treatment designed to reduce pain and improve mobility in patients with knee osteoarthritis.

Hymovis is a hyaluronic acid-based injection used to treat knee osteoarthritis, providing lubrication and cushioning to reduce pain and improve mobility.

Clinical data indicate that Radiesse is a key competitor within the anti-aging treatment market. Read more on Doctor Medica.
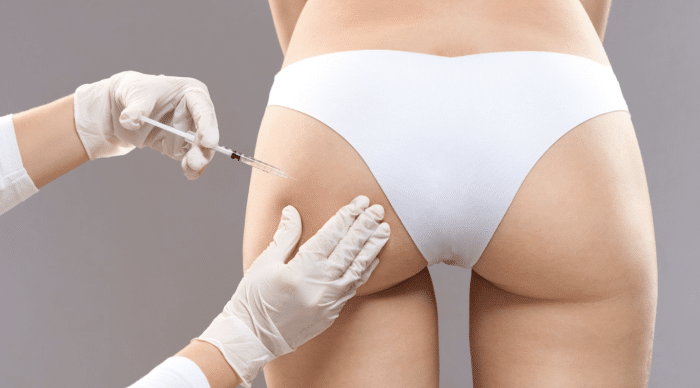
The Radiesse butt lift procedure involves stimulating collagen production to achieve a natural-looking enhancement.

Radiesse is a dermal filler that can add volume and contour to the jawline, providing a more sculpted and youthful appearance.

Radiesse and Juvederm use different materials to smooth wrinkles. Read more on Doctor Medica.

Clinical data supports the efficacy of Radiesse for cheek enhancement, showcasing long-term results and high patient satisfaction rates.
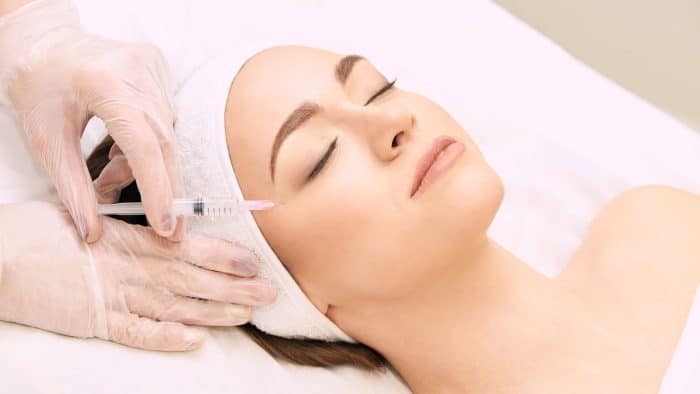
Common side effects of Radiesse include swelling, bruising, and redness, which are typically mild and resolve within a few days.

Radiesse is a dermal filler that can effectively rejuvenate aging hands by restoring volume and stimulating collagen production.

Radiesse contains calcium hydroxylapatite (CaHA), while Restylane is based on hyaluronic acid (HA). Read more on Doctor Medica.

Radiesse and Sculptra are both popular types of fillers used in cosmetic procedures, each offering unique benefits for facial rejuvenation.